Ensuring GMP and MDR compliance through better contract data and playbooks
Life sciences and healthcare companies operate under strict regulatory requirements, including the EU’s Good Manufacturing Practice (GMP) guidelines and the Medical Device Regulation (MDR) (Regulation (EU) 2017/745). Demonstrating compliance increasingly relies on clearly defined responsibilities, traceable documentation, and well-managed contractual relationships
Structured contract data management and standardized playbooks help organizations meet these expectations. By clearly defining roles, responsibilities, and oversight processes, contracts become an important tool for maintaining regulatory compliance and supporting audits.
This article will cover:
- How contracts support key GMP compliance requirements
- Why MDR compliance depends on disciplined contract controls
- The role of contract playbook templates in medical device regulation
- How CLM platforms improve traceability and audit readiness
Key GMP compliance requirements via contracts
GMP guidelines require Marketing Authorization Holders (MAHs), Manufacturing Importation Authorization (MIA) holders, and contract manufacturers to have written agreements in place whenever manufacturing or quality-related activities are outsourced. In certain setups, these agreements can form a “chain of contracts” linking multiple parties involved in the manufacturing process, however direct written contracts between the performing parties are generally preferred.
According to GMP Chapters 4 and 7, these contracts should address these key elements:
- Clear communication and documentation procedures between the parties, including access to relevant contractual and quality records
- Written assessment by the MIA holder’s Qualified Person (QP) confirming that the arrangements are suitable and support GMP compliance
- Integration of contractual responsibilities into the Pharmaceutical Quality System (PQS), including consideration in Product Quality Reviews (PQRs)
- Review of records from outsourced activities by QP, while the MIA holder remains ultimately responsible for GMP compliance
Quality or Technical Agreements (QAs/TAs) are central to this setup. Unlike commercial contracts, they focus specifically on GMP responsibilities and oversight for outsourced activities. These agreements should clearly describe who is responsible for what, how oversight works, and how the arrangement fits into the overall GMP system.
MDR compliance for contract manufacturers
EU MDR expands the compliance responsibilities for contract manufacturers. They’re expected to run well-structured processes and maintain solid documentation within their quality management system (QMS). Their contracts should clearly spell out these responsibilities so companies remain audit-ready and products stay fully traceable.
Key requirements under MDR and related standards such as ISO 13485 typically include:
- Documented design controls
- Batch records and deviation management
- Standard operating procedures (SOPs)
- Personnel training records
- Oversight of the supply chain
For highly regulated products, agreements should also address widely recognized data integrity principles such as ALCOA+, the management of electronic batch records, and requirements for full traceability of medical devices throughout the product lifecycle.
The role of contract data and playbooks
CLM platforms help organizations manage contracts more systematically while supporting compliance with EU GMP and MDR requirements.
For legal and QA teams, this provides clearer oversight of contractual obligations and stronger control over how responsibilities are defined and tracked. Common CLM capabilities that support this include:
- Audit trails and version histories that support regulatory inspections
- Pre-approved playbook templates for QAs/TAs, master service agreements (MSAs), and supply contracts to maintain regulatory alignment
- Contract data extraction tools that help verify roles, responsibilities, and changes across the supply chain
- Integrations with quality systems, such as Veeva Vault or eTMF, to centralize documentation and improve QMS data flows
Well-designed contract playbooks further support regulatory compliance. They often include clauses and checklists that address key regulatory requirements, such as:
- GMP and MDR responsibility clauses for outsourced activities
- Product lifecycle stage provisions, covering development, manufacturing, and distribution
- Jurisdiction-specific provisions, particularly for Civil Law systems common in EU cross-border operations and corporate group structures
Playbooks can also incorporate requirements from GDPR and EUDRALEX Annex 11, helping ensure that electronic records meet expectations for both data privacy and data integrity.
While CLM systems and contract playbooks are not mandated by GMP or MDR, they can provide the structured documentation and traceability that inspectors expect to see in practice.
Read next: A comprehensive guide to today’s CLM
🔑 Key takeaways
- GMP and MDR compliance depend on clear, well-structured contracts across the supply chain.
- Standardized contract templates and playbooks help ensure that key regulatory clauses, such as GMP responsibilities, lifecycle requirements, and jurisdictional provisions, are consistently included.
- CLM platforms provide audit trails, version histories, and centralized contract data that help organizations maintain compliance and prepare for regulatory inspections.
FAQs
Contracts define the responsibilities between parties involved in outsourced manufacturing or quality activities. Under EU GMP Chapter 7, written agreements must clearly outline roles, communication procedures, and quality oversight to ensure regulatory compliance.
The EU Medical Device Regulation expands compliance responsibilities for contract manufacturers, requiring structured processes, documented quality controls, and full traceability of devices throughout the product lifecycle.
Contract playbooks provide standardized clauses and templates that help organizations consistently include key regulatory requirements such as GMP responsibilities, MDR traceability provisions, and data integrity standards.
CLM platforms centralize contract data, maintain version histories, and provide audit trails. This makes it easier to demonstrate compliance, review contractual obligations, and prepare documentation during regulatory inspections.
Compare European CLM leaders
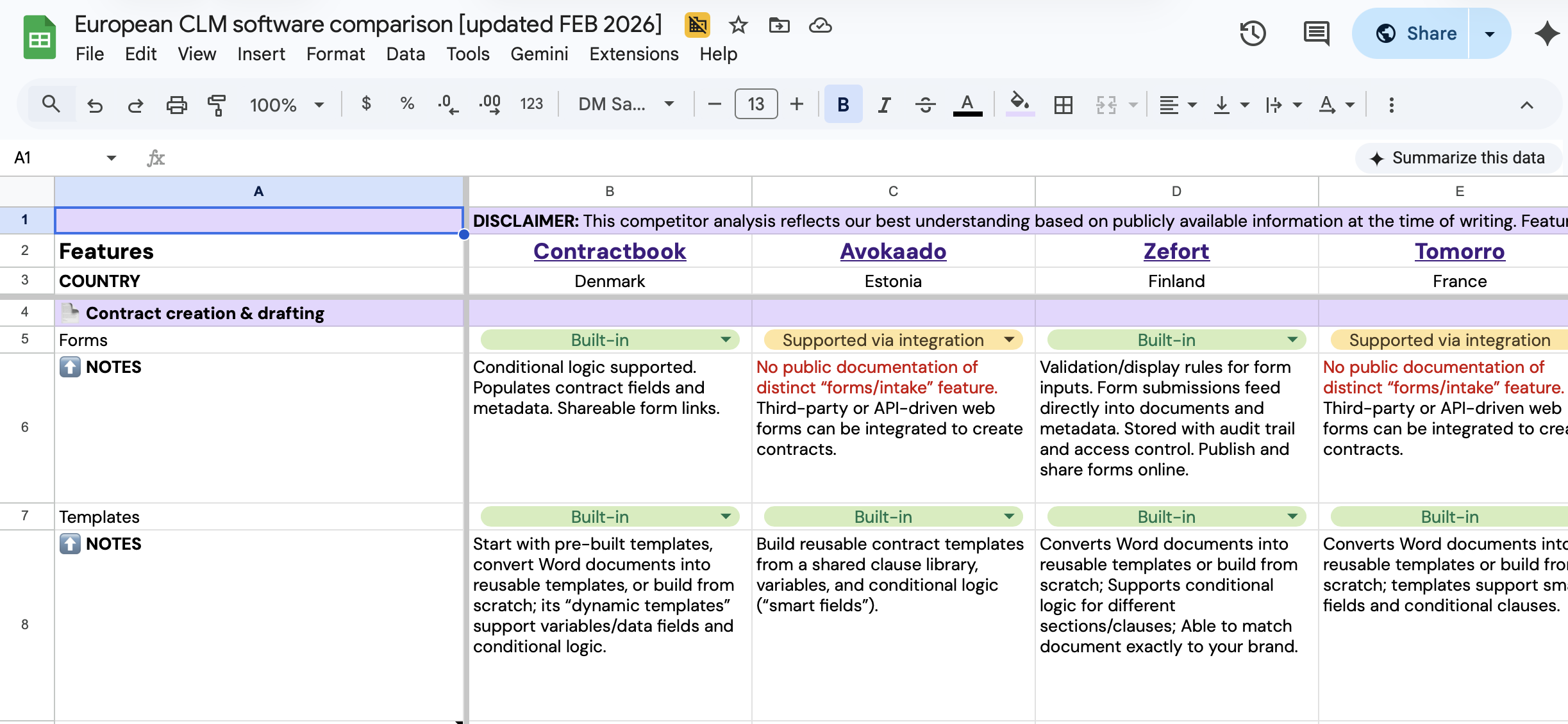
Get a comprehensive breakdown of the top CLM solutions in one spreadsheet.
Compare European CLM leaders

Get a comprehensive breakdown of the top CLM solutions in one spreadsheet.




