Contract management for biotech: protecting IP across hundreds of clinical trials
Protecting intellectual property (IP) in biotech contract management requires precision. With hundreds of clinical trials and volume partnerships, biotech firms face unique IP compliance risks that amplify regulatory, financial, and reputational exposure. The stakes are high: clear contract terms, robust trade secret protections, and scalable contract lifecycle management (CLM) tools are all crucial for success.
This article will explore:
- Key contract mechanisms to secure IP in biotech research, development, and licensing
- Tactics for managing hundreds of agreements while safeguarding trade secrets and data
- How scalable CLM tools reduce regulatory, financial, and reputational risks
- Real-world compliance considerations in clinical trials and technology transfer
Core IP protection mechanisms in biotech contracts
In contract management for biotech, detailed agreements define who owns which innovations. Most biotech trials involve external partners, such CROs, universities, and suppliers, making it essential to specify IP terms clearly. Here are the main protection methods:
1. Patents as the foundation
Patents provide up to 20 years of protection for core inventions such as molecules, formulations, or manufacturing platforms, typically calculated from the filing date. In the EU, Supplementary Protection Certificates (SPCs) can add up to five years, with a possible six-month pediatric extension, aiming to deliver up to around 15 years of effective protection from first EEA marketing authorization.
In parallel, EU regulatory data protection has traditionally followed the “8+2(+1)” model: eight years of data exclusivity, two years of market exclusivity, and a potential additional year for a new indication with significant clinical benefit. Under the EU Pharma Package, this is shifting to an “8+1(+1+1)” structure, with additional exclusivity tied to meeting policy objectives such as timely EU launch or addressing unmet medical need.
Patents require public disclosure and remain central to licensing and partnering, while regulatory exclusivities provide a distinct, increasingly policy-driven layer of protection.
2. Trade secrets for unpatentable assets
Trade secrets are a key tool for protecting valuable assets that cannot, or should not, be patented, such as confidential know-how, manufacturing processes, clinical data, algorithms, and development strategies. Under the EU Trade Secrets Directive (2016/943), protection arises where information is secret, has commercial value because it is secret, and is subject to reasonable steps to keep it confidential. The UK provides a parallel regime under the Trade Secrets (Enforcement, etc.) Regulations 2018.
In practice, maintaining protection depends on robust contractual and organizational measures. Companies typically rely on NDAs, employee IP and confidentiality assignments, and carefully drafted confidentiality and use restrictions in collaboration and services agreements to demonstrate that “reasonable steps” have been taken.
Unlike patents, trade secrets can last indefinitely, but only for as long as secrecy is preserved. EU law allows companies to take action if someone steals or misuses their trade secrets. However, it does not stop others from legally reverse engineering a product or developing the same idea on their own, unless a contract specifically prohibits this.
3. Hybrid agreements
R&D collaborations often combine research services, joint development, and IP licensing in a single hybrid structure. Contracts must spell out who may do what with the IP, distinguishing background IP (pre‑existing technology) from foreground IP (new results, data, and inventions generated in the project), and clearly allocating ownership, access rights, and commercialization paths.
Typical issues include whether licences are exclusive or non‑exclusive, how and when parties can publish results, and which party controls patent filing and enforcement for foreground inventions. It is common to define option periods for future licences and clear negotiation windows before any enabling public disclosure, ensuring patent filings can be made in time.
Overview of IP protection mechanisms
The following table outlines different IP protection mechanisms in biotech contracts and how they tend to apply in clinical trials and development work.
| Mechanism | Strengths | Trade‑offs | Typical use in clinical trials |
|---|---|---|---|
| Patents | Enforceable exclusivity; attracts investors; licensable | Public disclosure; high cost and complexity | Core drug substances, formulations, key methods |
| Trade secrets | No disclosure required; potentially indefinite duration | Vulnerable to reverse engineering and leaks | Protocols, manufacturing know‑how, raw clinical data |
| Regulatory data exclusivity | Protects marketing authorisation dossiers from reliance; complements patents | Limited to regulatory contexts; time‑limited | Reference product clinical dossiers used in MA filings |
| Licensing/collaboration | Access to external resources; risk‑sharing; flexibility | Complex allocation of rights and control; negotiation costs | Partnering with CROs, universities, and CMOs for R&D and trials |
Contract strategies for scale across hundreds of trials
Managing hundreds of clinical trial agreements makes IP and regulatory compliance a coordination challenge rather than a purely legal one. Standardizing templates is important, but each contract must still address project‑specific IP, data use, and regulatory risks:
- Front-load term sheets: Negotiate IP scope, payment triggers, commercialization rights, and research timelines early. This helps avoid later disputes.
- Background vs. foreground IP: Distinguish background IP (what each party brings) from foreground IP (what’s created). Address joint ownership, grant-backs, platform carve-outs, patent filing responsibility, costs, and revenue sharing.
- Risk and compliance clauses: Go beyond generic “good clinical practice” wording. Include clear audit rights, defined quality and compliance obligations, and references to the relevant regulatory frameworks (such as FDA, EMA, MHRA, or NHS/HRA requirements, as applicable). Allow time for patent filing before publications.
- University-industry deals: Offer pathways to exclusive licences while respecting publication and funding obligations. Clarify tech transfer roles, regulatory dossier ownership, and how data and materials transition from research to clinical development.
Implementing CLM for high-volume management
Manual oversight of hundreds of contracts increases the risk of missed obligations, inconsistent IP terms, and opaque risk exposure. A scalable CLM tool centralizes oversight and helps ensure compliance with regulatory and IP requirements. Key capabilities include:
- Automated obligation tracking: Track IP‑related clauses, reporting obligations, milestones, patent filing deadlines, and regulatory timelines linked to marketing authorization, data exclusivity, and SPC expiry, with automated alerts
- AI‑driven risk flagging: Use AI to automatically flag high‑risk clauses in R&D agreements, clinical trial agreements, manufacturing agreements, and data‑sharing contracts.
- Central contract repository: Maintain a secure, searchable repository with access controls, version management, and audit trails, enabling legal and compliance teams to demonstrate consistent use of approved wording and to support audits.
- Cross‑functional workflows: Integrate legal, business, regulatory, and scientific stakeholders into approval workflows for template updates, onboarding of new partners, amendments, and renewals, so that IP, clinical operations, and compliance constraints are considered together
Applied effectively, CLM platforms have been shown to improve visibility into risk and support adherence to regulatory deadlines, which can indirectly help shorten time to first‑in‑human studies and market launch while reducing the likelihood of costly compliance failures.
Read next: 10 best contract management software in 2026
Regulatory and enforcement considerations
Biotech IP sits within a complex global and EU regulatory framework. The WTO Agreement on Trade-Related Aspects of Intellectual Property Rights (TRIPS) sets baseline standards for patents and trade secrets, while EU regimes, such as SPCs, data protection, and orphan drug exclusivity, can extend market protection for qualifying products.
EU orphan designation typically grants 10 years of exclusivity (up to 12 with pediatric compliance), though reforms may adjust these periods. This makes IP due diligence in pharma and biotech M&A critical, including review of patent scope, exclusivity status, licensing constraints, and enforceability in key markets.
Protection also depends on operational discipline: strong NDAs, clear confidentiality terms, robust information security, and clear allocation of regulatory and IP responsibilities in manufacturing agreements. With regulators imposing substantial fines for compliance failures, IP-aware contracting and structured contract management are essential to protect value and reduce enforcement risk.
🔑 Key takeaways
- IP protection in biotech is multi-layered. Patents, trade secrets, regulatory data exclusivity, and contractual controls all work together to protect innovation.
- Clarity in contracts is critical. Agreements must clearly define background vs. foreground IP, licensing rights, publication rules, patent control, and regulatory responsibilities.
- Scaling trials increases coordination risk. Standardized templates, front-loaded term sheets, and consistent risk clauses are essential.
- CLM helps reduce legal and regulatory exposure. Scalable CLM tools improve visibility, automate obligation tracking, flag risk, and help prevent costly compliance failures or missed exclusivity deadlines.
FAQs
Companies use detailed contracts that clarify IP ownership, licensing rights, and background versus foreground IP. They rely on patents for core inventions, trade secrets for confidential know-how, and versatile agreements to balance collaboration with clear IP rules, helping prevent disputes among CROs, universities, and suppliers.
Confidential information like protocols, datasets, or clinical know-how is protected through nondisclosure agreements, employee IP assignments, and strong confidentiality clauses. These safeguards can offer indefinite protection if the information remains secret.
Data integrity failures, Good Manufacturing Practice (GMP) violations, confidentiality breaches, and unclear IP ownership can trigger regulatory penalties, litigation, lost exclusivity, and reputational damage.
Standard templates help ensure consistent handling of IP, compliance, and risk terms across all agreements. However, customization is necessary for project-specific risks, ownership terms, and regulatory concerns in each trial or partnership.
CLM platforms automate obligation tracking, flag high-risk clauses, and centralize all contracts for consistent access control and compliance. They help biotech firms manage large volumes of agreements efficiently, reducing manual errors and supporting regulatory and IP requirements across global partnerships.
Compare European CLM leaders
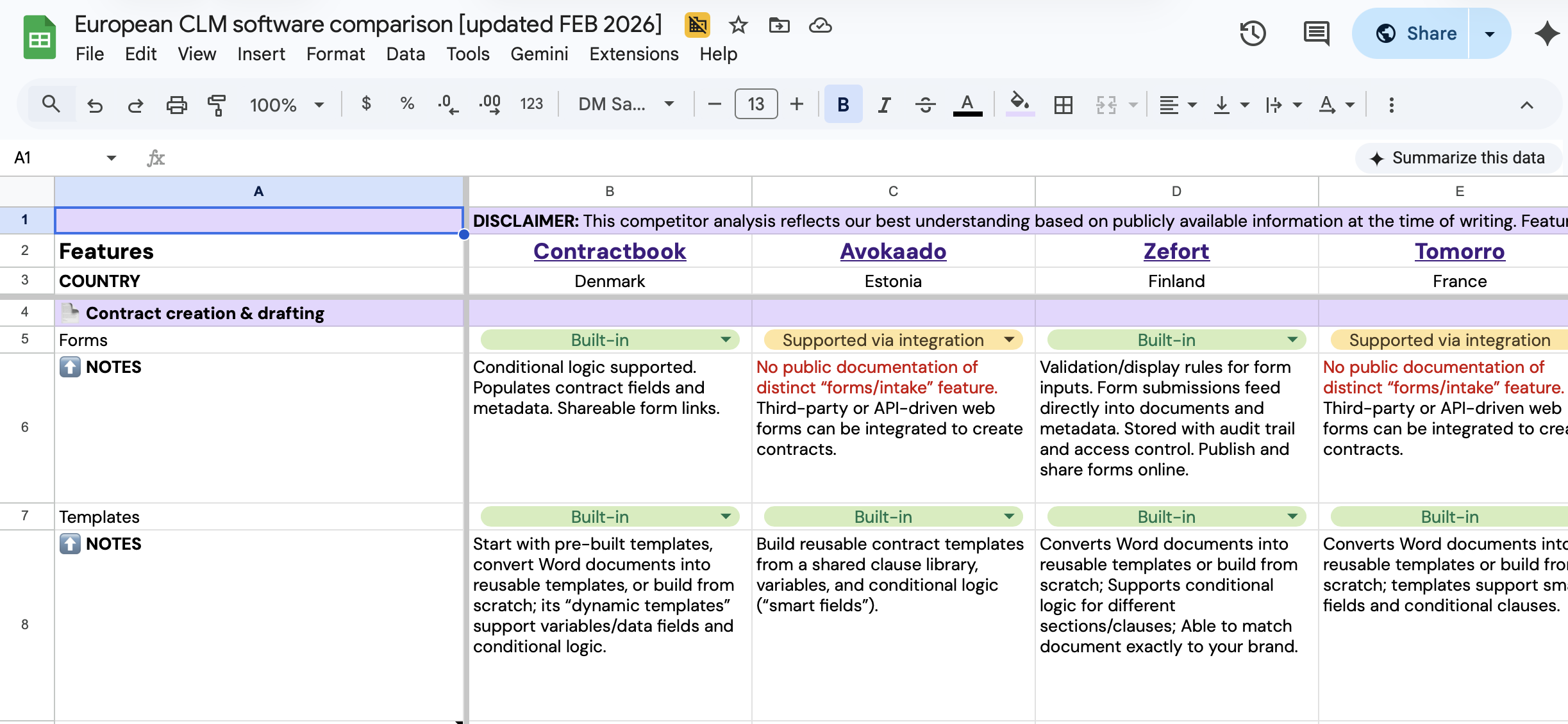
Get a comprehensive breakdown of the top CLM solutions in one spreadsheet.
Compare European CLM leaders

Get a comprehensive breakdown of the top CLM solutions in one spreadsheet.



